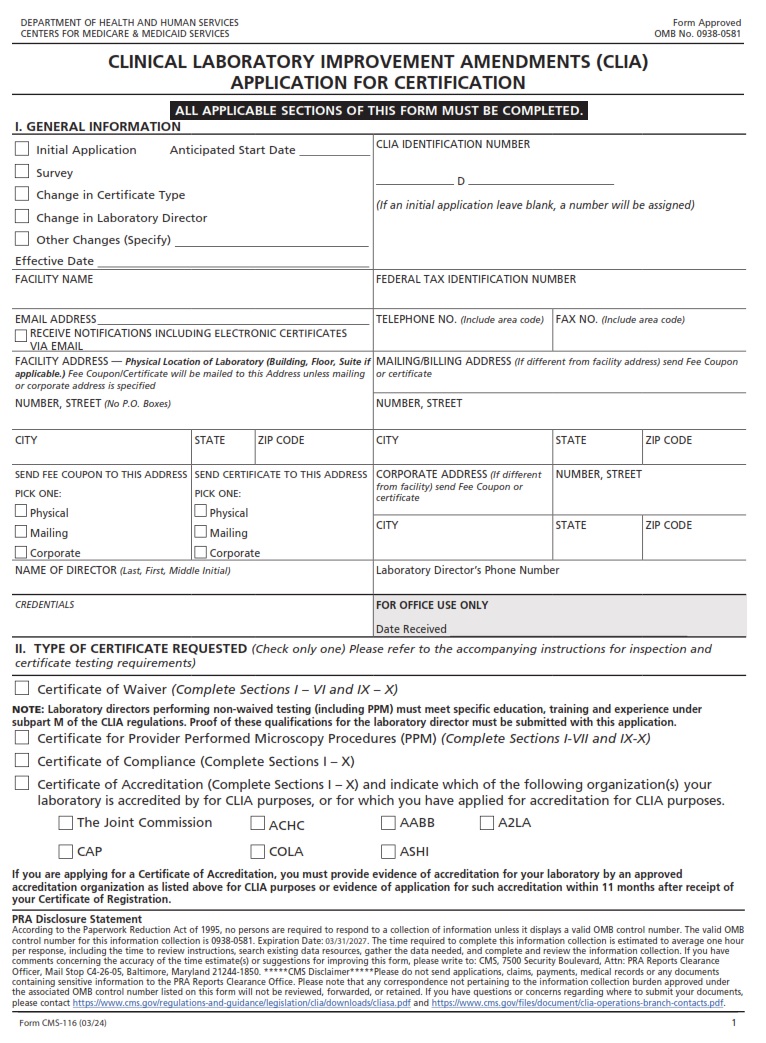
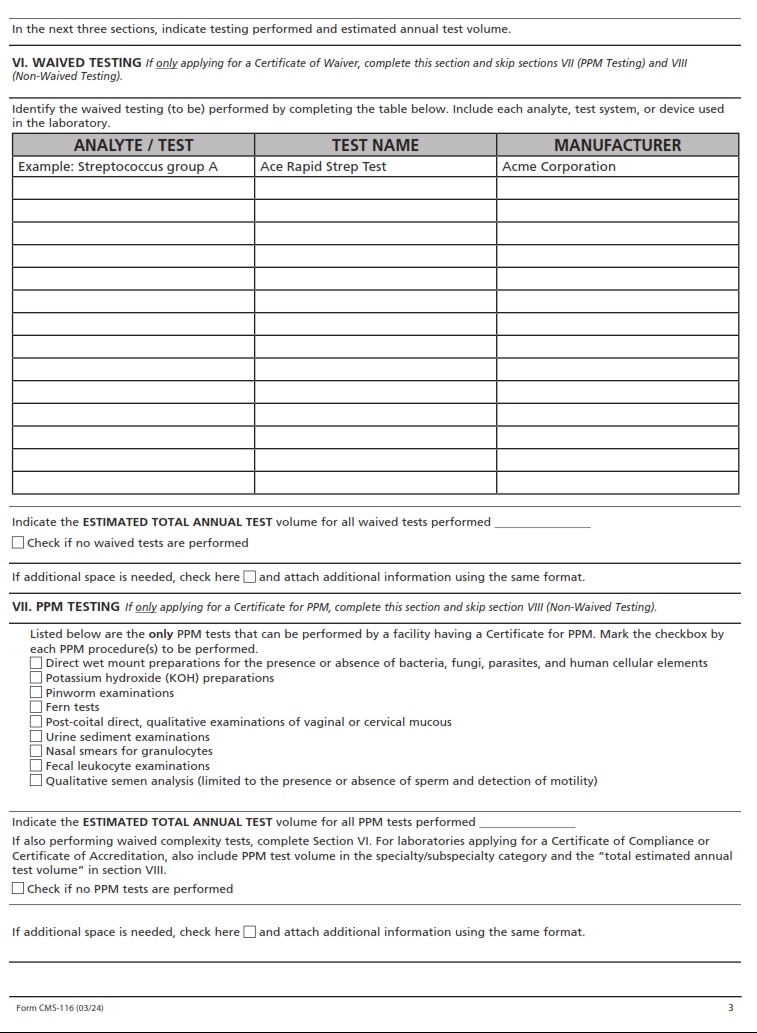
CMSFORM.ORG – CMS 2802C – REQUEST FOR VALIDATION OF ACCREDITATION SURVEY FOR HOME HEALTH AGENCY – As the healthcare landscape continues to evolve, ensuring the quality and reliability of services provided by home health agencies becomes paramount. In this digital era, where data reigns supreme, the Centers for Medicare & Medicaid Services (CMS) has introduced the form CMS 2802C – Request for Validation of Accreditation Survey for Home Health Agency to streamline and enhance accreditation processes. This document serves as a critical tool in evaluating the performance and adherence of home health agencies to established standards, ultimately safeguarding the well-being of patients and promoting excellence in care delivery. Join us as we delve into the intricacies of this validation process, unraveling its significance in shaping the future of home healthcare services.
Download CMS 2802C – REQUEST FOR VALIDATION OF ACCREDITATION SURVEY FOR HOME HEALTH AGENCY
[su_table responsive=”yes”]
| Form Number | CMS 2802C |
| Form Title | REQUEST FOR VALIDATION OF ACCREDITATION SURVEY FOR HOME HEALTH AGENCY |
| Published | 2019-03-31 |
| O.M.B. | – |
| File Size | 57 KB |
[/su_table]
[su_button url=”https://cmsforms.org/wp-content/uploads/2024/12/CMS-2802C-REQUEST-FOR-VALIDATION-OF-ACCREDITATION-SURVEY-FOR-HOME-HEALTH-AGENCY.pdf” target=”blank” background=”#8ec54f” size=”7″ center=”yes” icon=”icon: file-pdf-o” title=”Download CMS 2802C”]Download CMS 2802C [.PDF][/su_button]
What is a CMS 2802C?
The CMS 2802C, also known as the Request for Validation of Accreditation Survey for Home Health Agency, is a crucial document in the healthcare industry. It serves as a means for home health agencies to validate and demonstrate their compliance with accreditation standards set by the Centers for Medicare & Medicaid Services (CMS). Through this survey, agencies are able to showcase their commitment to providing quality care and meeting regulatory requirements.
This validation process is essential for ensuring that home health agencies deliver safe and effective services to patients. By undergoing accreditation surveys using the CMS 2802C form, these agencies can identify areas for improvement and implement best practices in patient care. Ultimately, this helps in enhancing transparency, accountability, and overall quality of care within the home health sector.
Where Can I Find a CMS 2802C?
If you’re on the hunt for a CMS 2802C form to request validation of accreditation survey for your home health agency, there are several avenues you can explore. One option is to visit the official Centers for Medicare & Medicaid Services (CMS) website. Here, you may be able to download a digital copy of the form or find information on where to obtain a physical version.
Another valuable resource is reaching out to your state’s department of health or local healthcare regulatory agencies. They often have copies of important forms like the CMS 2802C available for providers in their jurisdiction. Additionally, networking with other home health agencies or industry professionals can lead you to sources where these forms are readily accessible. Remember, staying informed and proactive in seeking out such documentation is crucial in maintaining compliance and quality care standards within your agency.
CMS 2802C – REQUEST FOR VALIDATION OF ACCREDITATION SURVEY FOR HOME HEALTH AGENCY
Navigating the intricate world of healthcare accreditation can be a daunting task for home health agencies. The CMS 2802C form, requesting validation of accreditation survey, is a crucial step in ensuring compliance and quality care delivery. This process serves as a checkpoint to evaluate the agency’s adherence to established standards and regulations.
For home health agencies, undergoing accreditation surveys is not just about meeting requirements but also about upholding their commitment to providing exceptional care. The CMS 2802C form signifies the agency’s dedication to continuous improvement and patient safety, showcasing a proactive approach towards enhancing healthcare services. Embracing this validation process as an opportunity for growth can lead to increased trust from patients, improved outcomes, and a competitive edge in the ever-evolving healthcare landscape.
CMS 2802C Example